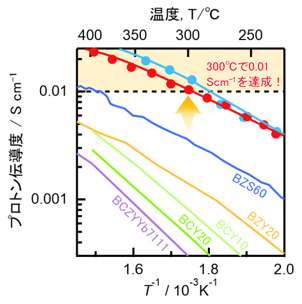
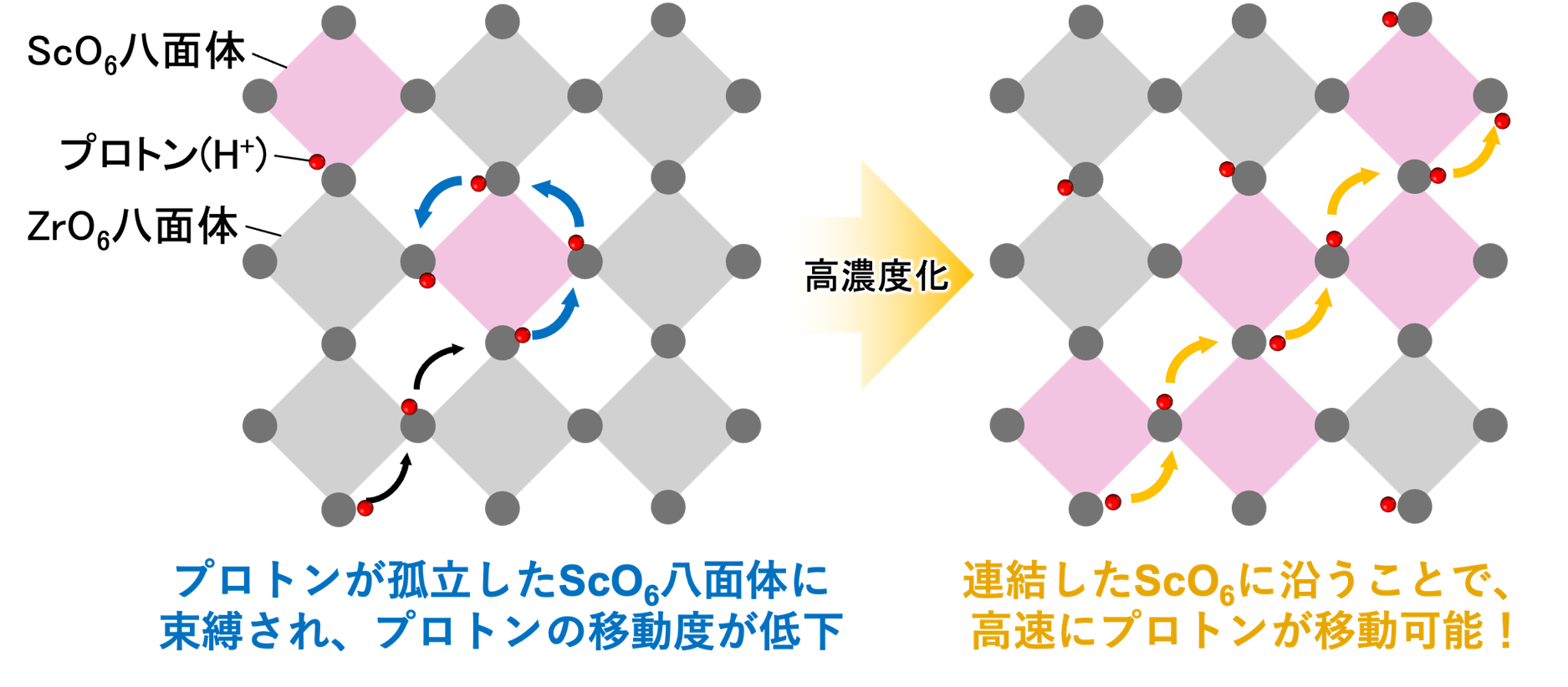
Solid oxide fuel cells (SOFCs) are among the most efficient and durable types of fuel cells. They use hydrogen as a fuel and generate electricity without emitting carbon dioxide during operation, and therefore have attracted considerable attention as a key technology for realizing a hydrogen-based energy society. However, SOFCs typically operate at high temperatures of 700–800°C, and the material cost associated with the use of expensive heat-resistant components remains a major challenge. If electricity generation could be achieved in the intermediate-temperature range of around 300°C, costs could be reduced by enabling the use of more affordable heat-resistant materials. To date, however, no electrolytes with sufficient performance in this temperature range had been identified. Our research group has developed an oxide that satisfies a key requirement for SOFC electrolytes—namely, a proton conductivity more than 0.01 S cm⁻¹—at 300°C by introducing high-concentration scandium (Sc) substitution into barium stannate (BaSnO₃) and barium titanate (BaTiO₃) (Fig. 1). In addition, through a collaborative study with domestic researchers specializing in computational science and electron microscopy, we investigated the proton conduction mechanism and found that a distinctive atomic arrangement consisting of connected ScO₆ octahedra enables rapid proton transport within the crystal (Fig. 2). It is the first to propose and experimentally demonstrate a method to avoid proton trapping, which has impeded performance improvements in proton-conducting oxides since their discovery in 1981. Our findings are expected to contribute to the realization of low-cost, intermediate-temperature SOFCs and to greatly accelerate the practical implementation and diversification of SOFC technologies.